 |
|
 |
| Shared Idea |
 |
| Waves
|
| Everything is either matter (solids, liquids and gases) or energy. Waves are a form of transferring energy. There are various types of waves, but three are most likely present in your immediate vicinity. They are mechanical waves, electromagnetic waves and sound waves.
|
| Waves also can be grouped as one of two types: either transverse or longitudinal. In a transverse wave, the wave oscillates at right angles to the direction of its advancement.
In a longitudinal wave, the wave oscillates parallel to the direction of its advancement. A simplistic way of picturing this is that the wave is moving up and down as it advances in a transverse wave. It moves back and forth as it advances in a longitudinal wave.
|
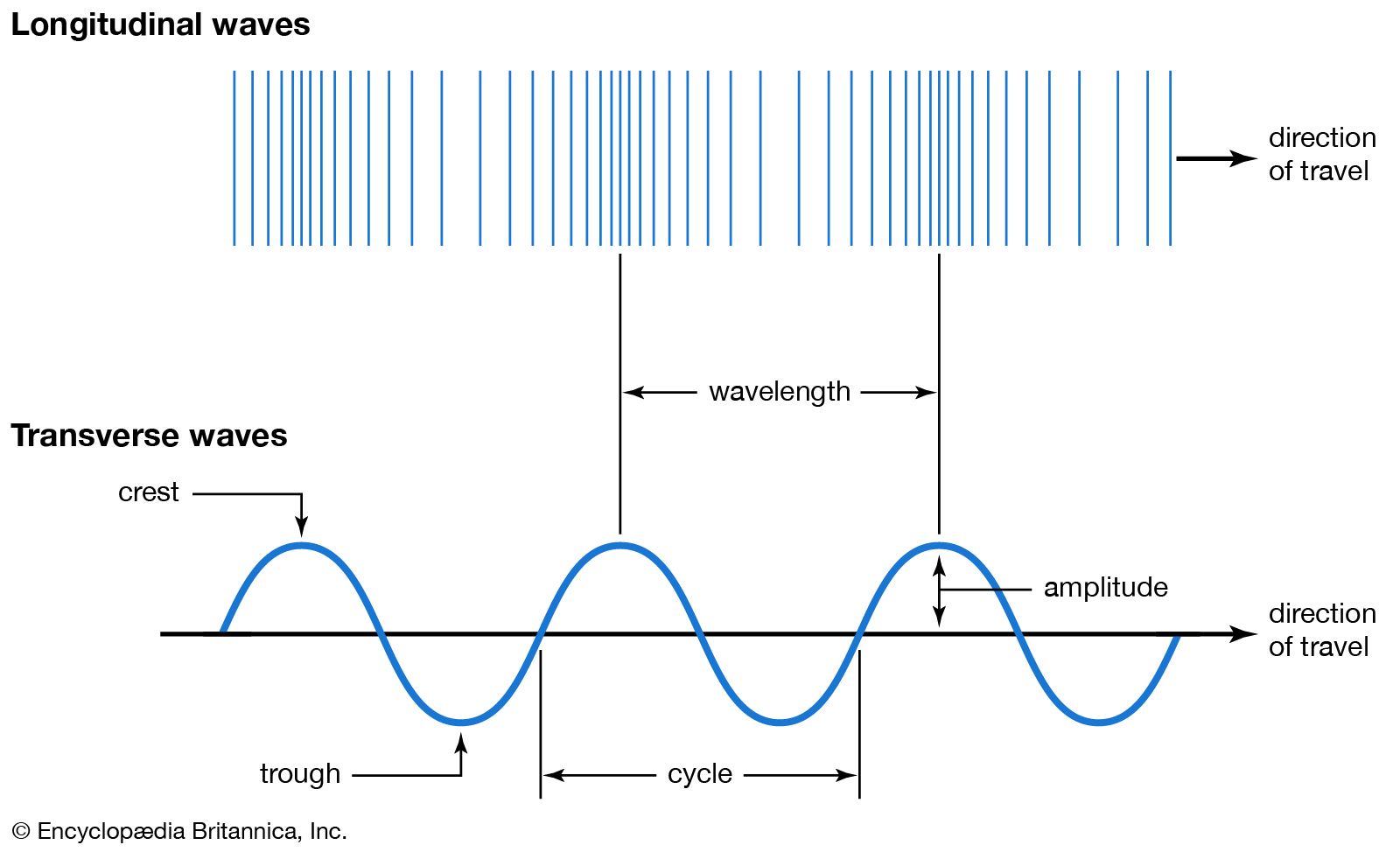 |
| A slinkie produces mechanical waves and can be used to visualize these two forms of waves. Lay the slinkie on a flat surface, such as the floor or a clear table. If the slinkie is stretched and one end is held stable, a transverse wave is produced when the other end is moved at right angles. A longitudinal wave is produced when the other end is moved back and forth. The resulting waves are influenced by how much energy is exerted into the moving end. It also helps if the energy is exerted in a steady pattern.
|
| There are seven types of electromagnetic waves. They are radio waves, microwaves, infrared light, visible light, ultraviolet light, x-rays and gamma rays. They vary according to the energy and wavelength, which are inversely proportional. That is, as one increases, the other decreases. The ratio between these factors is known as Planck's constant. The colors of the rainbow reflect these factors.
|
| Why is the sky blue? This classical question can lead to a variety of responses, but the correct answer reflects the visible light portion of the electromagnetic spectrum. As light moves through a medium, it is bent or diffracted by particles with a size that is equal to or greater than the light's wavelength. Purple and blue have the shortest wavelengths. The small particles in the atmosphere scatter this light, creating the blue sky that we therefore observe. When light is passing through the atmosphere at a lower angle, the purple and blue light has already been scattered. The larger particles, which are at a lower altitude, now scatter the orange and red end to the spectrum. This is why the sky has those hues at sunrise and sunset.
|
| What happens when all of the visible light is scattered at the same time? If you add all of the colors in light (not paint), the result is the color we discern as white. Examples include milk, mayonnaise, cream cheese and clouds. This is even true for thick clouds, but they are only seen as white if observed from above. When we are beneath such clouds, they appear as grey because the colors of light have already been removed. When we look at a surface, such as clothing or paintings, we are seeing the light that has not been absorbed. That means that the actual surface is the opposite color of the one we see!
|
| Sound waves are different from light waves or most mechanical waves, such as ocean waves. They are longitudinal. A sound wave causes particles to move back and forth. This is why sound cannot pass through a vacuum. (In bygone days, it was believed that a celestial ether filled space, making it possible for electromagnetic waves to be transferred.) If you strike a tuning fork, you will hear the resulting sound. The pitch of that sound depends on the length and structure of the tuning fork. It may be difficult to perceive that sound is an actual longitudinal wave of particles in the air. This can be resolved if you can obtain an air vortex, such as an AirZooka. Pulling back the elastic strap at one end creates a mechanical wave of air particles. When it exits, it forms a sound wave that can then be both heard and felt at a moderate distance.
|
|
|





